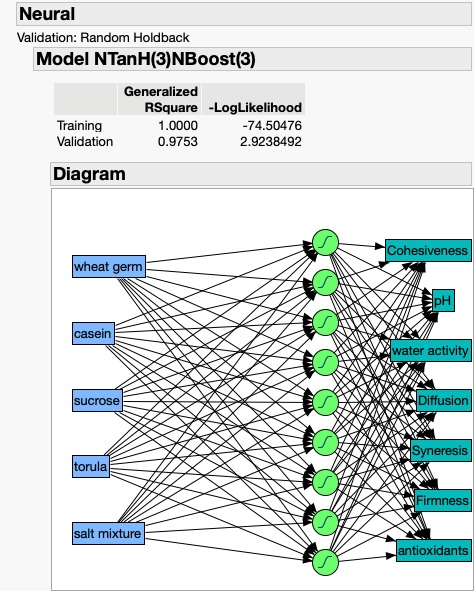
As an example of the potentially rich and productive field of exploration and discovery that can be reached through the integration of studies that tie the basic science and practical/applied aspects of insect rearing systems and their components, let me explain an example that I use in my insect rearing classes.
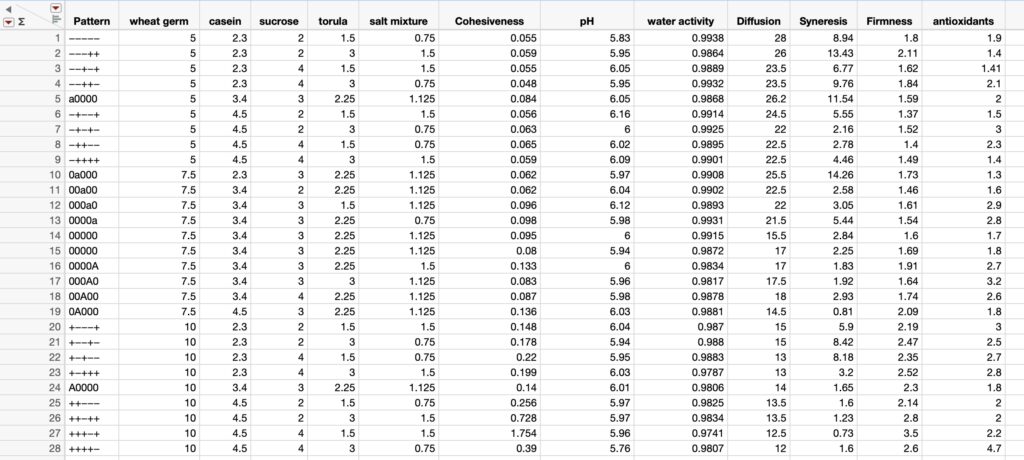
This narrative is based on an unpublished investigation that I did in my laboratory in recent years. I found a series of papers from the laboratories of Professors VandenBrooks and Harrison, including the paper by VandenBrooks et al. 2018 where the authors show that oxygen deficits for developing Drosophila melanogaster led to dramatic compensatory changes in the flies’ tracheal system and mitochondria. Though my background includes more 50 years of experience in physiological ecology, including studies of gas exchange, I was amazed at the profound effects that oxygen deprivation (holding developing larvae at about 10% O2) on the fundamental respiratory components of the flies. My amazement at the findings of this study led me to ask the question: how often do we find conditions in our rearing systems where the insects are being O2 -deprivation stressed (and likewise excess CO2 stressed)? I approached two of my colonies (painted lady butterfly larvae) with this question and with an instrument that measures headspace O2 and CO2, and I found a fairly small difference between rearing container gases and those in open laboratory air in my painted lady butterfly larvae cages; HOWEVER, in my waxworm cages I found that the O2 reading was about half that of laboratory air, and CO2 was about 180 times that of laboratory air (90,000 ppm vs. 500 ppm in lab air). The waxworm cages were modified Ziplock food storage containers with 24 pinholes in the lidding made with conventional dissecting needles (Figures 1 and 2).


Figure 1 (top), Ziplock container of waxworms (Galleria mellonella): container lid had 24 pinholes for gas exchange and Figure 2 (bottom), an O2 and CO2 headspace analyzer showing 10.7% O2 and CO2 at 9.0% or 90,000 ppm.
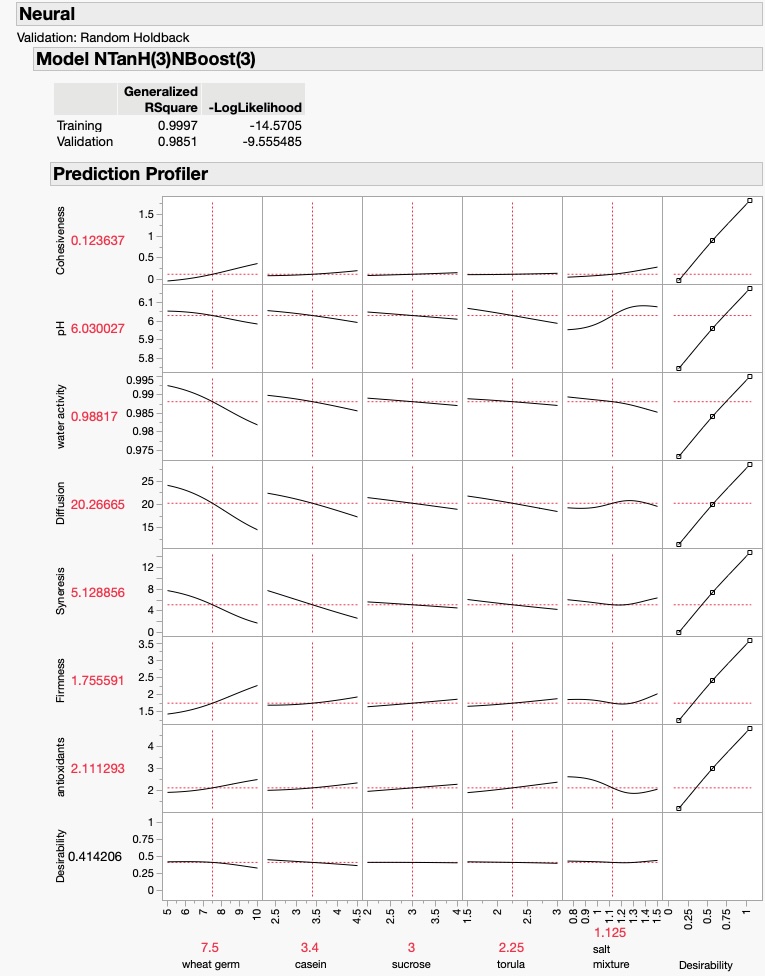
The first hypothesis that this question generated was that providing larger openings for gas exchange would improve the contents of O2 and CO2 and that in turn, a higher O2 and a lower CO2 would reduce stress and enhance the fitness of insects with improved gas exchange systems. I tested the first part of my hypothesis by placing 2 x 2 cm openings in the cage tops of the waxworm containers, replacing the cutout plastic with stainless steel mesh that was very porous (Figure 3). The comparison of the improvements can be seen in Figure 4, and preliminary observations of faster growth rates seemed to support the 2nd part of my hypothesis, but this hypothesis will require more testing.


The point of this narrative is that the dramatic findings from the VandenBrooks paper prompted me to do some deep thinking about how much we take for granted that our containers are doing their job of providing adequate oxygen and removal of excess carbon dioxide; but with insects that are densely packed and/or of high metabolic demands (such as waxworms), this lack of attention may be a basis for cryptically stressed insects.
This preliminary study is a good example of the kind of linking studies that the Special Topics Collection is striving to publish in order to help the rearing community appreciate the value of basic science studies of the biological and physical aspects of rearing systems and how these data-driven, science-based inquiries can lead to improvements in the rearing conditions in terms of food, gas exchange, environmental features, and all other aspects of reliable insect rearing systems.